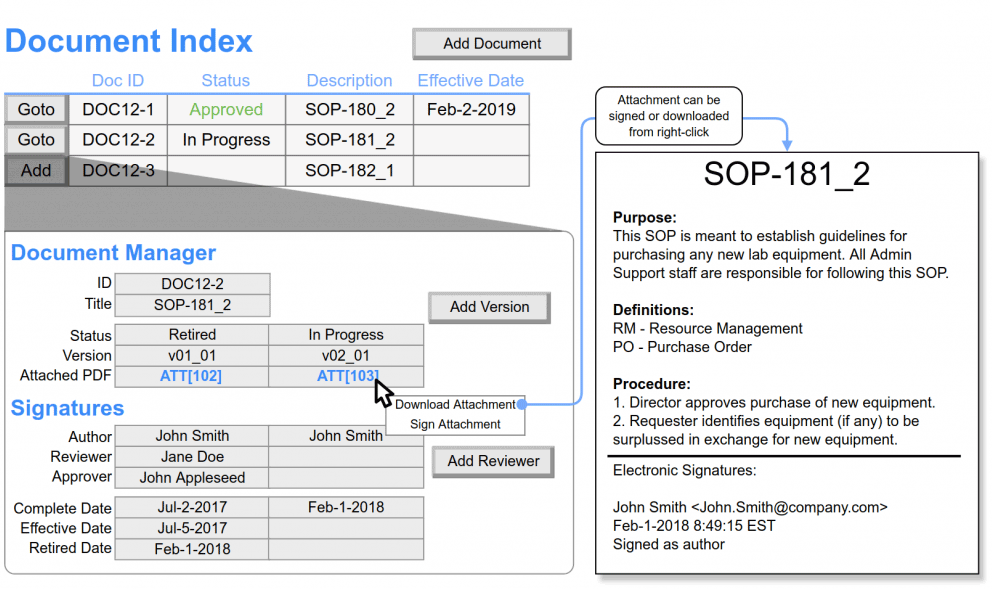
What is LIMS Software?
A Laboratory Information Management System (LIMS) is a software solution that streamlines and centralizes data management in a laboratory setting. It helps pharmaceutical laboratories maintain accurate records, improve data traceability, and ensure compliance with regulatory requirements.
Importance of Validation
The Importance of Validation in FDA-Compliant Laboratories
FDA regulations require pharmaceutical companies to validate their LIMS software to ensure its reliability, accuracy, and security. Validation serves as documented evidence that the software meets the specified requirements and operates consistently within the established parameters. Proper validation helps reduce the risk of data integrity issues and regulatory non-compliance.
Key Components
Key Components of Validation Packages for LIMS Software
Validation Packages can vary and include items such as a Validation Plan, Customer Administration Procedures, User Requirements Specification, Functional Requirements Specification, Design Qualification (DQ), Risk Assessment,
Operation Qualification (OQ) Vendor Audit Plan, Vendor Audit Report or Installation Qualification (IQ) or Summary Report, and a Configuration Specification document. Below are descriptions of some of the main components of a validation package.
- User Requirements Specification (URS): This is the foundational document that outlines the laboratory’s specific needs and requirements for LIMS. It serves as a reference point throughout the validation process.
- Functional Requirements Specification (FRS): The FRS details how the LIMS software will meet the requirements outlined in the URS. It defines the functionality of the system, such as sample tracking, data storage, and reporting.
- Installation Qualification (IQ): This phase ensures that the LIMS software is installed correctly and functions as expected in the laboratory environment. It involves hardware and software verification, and documentation is essential for compliance.
- Operational Qualification (OQ): OQ tests whether the software operates according to predefined specifications. This phase includes testing scenarios for functionality, security, and data integrity.
- Performance Qualification (PQ): PQ validates that the LIMS software consistently performs in a real laboratory environment. This involves testing under operational conditions to ensure that the system meets its intended use and regulatory requirements.
- Validation Summary Report: A comprehensive report summarizing the entire validation process, demonstrating that the LIMS software is suitable for its intended purpose and complies with FDA guidelines.
Choosing Validation
Choosing the Right Validation Package
When selecting a validation package for your LIMS software, it’s important to consider the specific needs of your laboratory. Look for a package that aligns with your URS and is designed to meet FDA compliance standards. Additionally, the package should offer ongoing support and updates to ensure continued compliance as regulations evolve.
Conclusion
Validation packages for LIMS software are a critical component of ensuring that pharmaceutical laboratories remain FDA compliant. By implementing a thorough validation process, laboratories can mitigate the risk of data inaccuracies, streamline their operations, and confidently meet regulatory requirements. Choose the right validation package to support the success of your laboratory’s quality control and data management processes.
SciCord
SciCord is a hybrid ELN/LIMS, featuring a spreadsheet paradigm, that combines the compliance and structured aspects of a Laboratory Information Management System (LIMS) with the flexibility of an Electronic Laboratory Notebook (ELN). The ELN and LIMS functions integrate seamlessly and enable your company to quickly reap the benefits of enhanced data and time efficiencies as you continue to meet compliance standards and follow GLP.