SciCord is attending the SERA-6 conference on Soil Testing in West Virginia, starting June 7th.
Register to meet us there!

temp text
SciCord is attending the SERA-6 conference on Soil Testing in West Virginia, starting June 7th.
Register to meet us there!

temp text
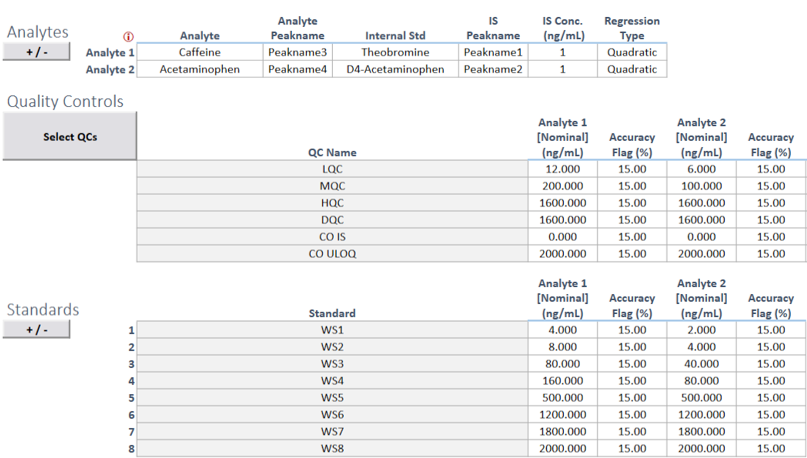

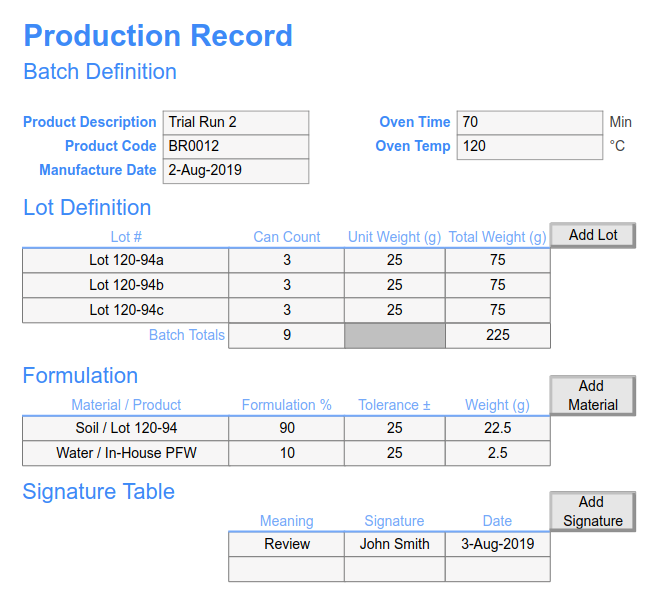
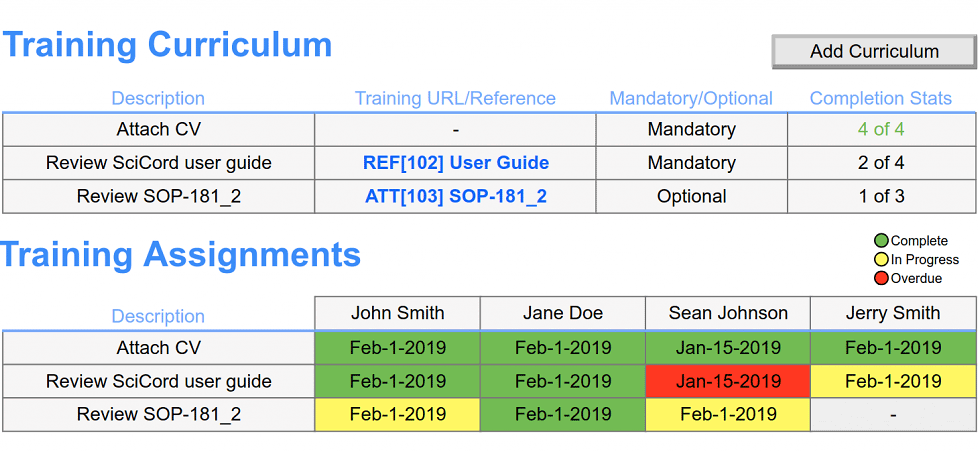
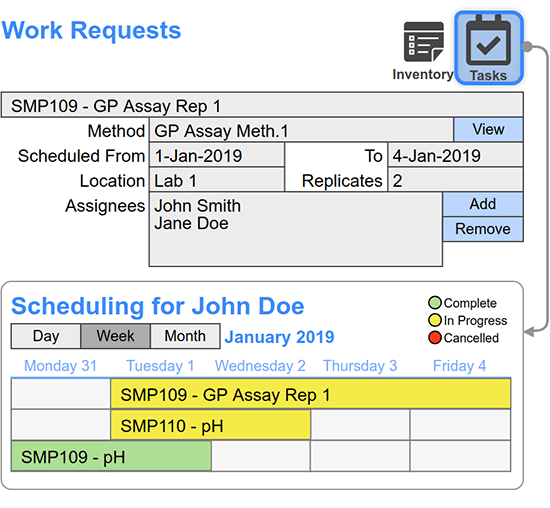
Reach out to Schedule a Meeting and get more information about how SciCord can fit into your lab
Don’t take our word for it.
We exceed our client’s demands everyday to make their research and discovery process simpler and more efficient.
This is by far the best value in science software (or anything else in science, really) that we’ve ever experienced. Other solutions in this price range had a fraction of the features, and those with the features cost 3x – 10x more. We’re very happy customers.

Josh Guyer,
Senior Pharmaceutical Scientist