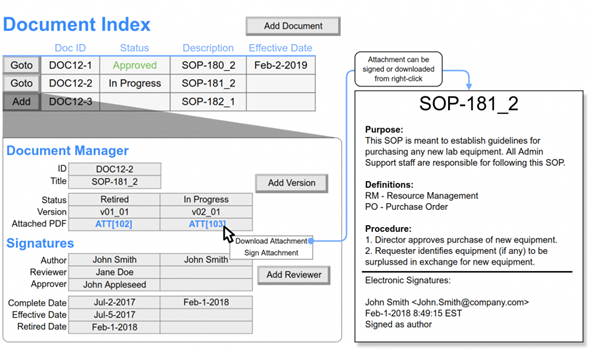
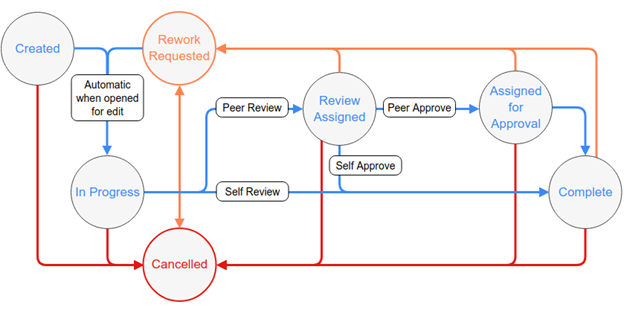
Platform
Our Lab Informatics platform combines the utility of a LIMS and versatility of an ELN
Top Features
Solutions
Looking for something more specific? Read through the other solutions we support out of the box
Resources
Browse our archive of case studies, functional documentation, and announcements.
Looking for something specific? Search our site:
temp text